ABSTRACT
Understanding surface passivation arising from aluminium oxide (Al2O3) films is of significant relevance for silicon-based solar cells and devices that require negligible surface recombination. This study aims to understand the competing bulk and surface lifetime effects which occur during the activation of atomic layer deposited Al2O3. We demonstrate that maximum passivation is achieved on n- and p-type silicon with activation at ~ 450 ◦C, irrespective of annealing ambient. Upon stripping the Al2O3 films and re-passivating the surface using a superacid-based technique, we find the bulk lifetime of float-zone and Czochralski silicon wafers degrade at annealing temperatures > 450 ℃. By accounting for this bulk lifetime degradation, we demonstrate that the chemical passivation component associated with Al2O3 remains stable at activation temperatures of 450─500 ℃, achieving an SRV of < 1 cm/s on n- and p-type silicon. In conjunction with the thermal stability, we show that films in the range of 3–30 nm maintain an SRV of < 1 cm/s when annealed at 450 ℃. From atomic-level energy dispersive X-ray analysis, we demonstrate that, post deposition, the interface has a structure of Si/SiO2/Al2O3. After activation at > 300 ◦C, the interface becomes Si/SixAlyO2/Al2O3 due to diffusion of aluminium into the thin silicon oxide layer.
1. Introduction
Silicon photovoltaics account for > 95 % of the PV market, and this dominance is predicted to remain unchanged for the foreseeable future . With the mainstream PV technology being the passivated emitter and rear cell (PERC) architecture, mitigation of surface recombination through state-of-the-art passivation layers on the front and rear of the solar cell has become ever more important. In particular, the reemergence of aluminium oxide (Al2O3) in 2006 has enabled high levels of surface passivation to be achieved on the rear side of PERC solar cells (on a p-type substrate), which can be attributed to the high levels of negative charge and good chemical passivation properties of the film . Furthermore, with the recent introduction of passivating contact structures (e.g. TOPCon), Al2O3 passivation is now being utilised on the front surface of TOPCon solar cells which use n-type substrates . As such, Al2O3 is playing an ever-increasing role in mitigating surface recombination on either the front or rear surface, thereby demonstrating its significance in the development of highly efficient solar cells.
There remains some uncertainty in how one can maximise the highest level of surface passivation on silicon. Often, the level of surface passivation is quantified based on the effective lifetime level achieved, which is subsequently converted into a surface recombination velocity (SRV) or surface saturation current density (J0s). However, in doing so, the underlying bulk silicon material is assumed to be thermally stable at post-deposition annealing temperatures of < 500 ℃. If this assumption is incorrect there can be substantial variability in the extracted values and thus any conclusions drawn from them. It has recently been shown that the bulk lifetime (τbulk) of float-zone (FZ) silicon is thermally unstable at temperatures between 400─800 ℃, whereby τbulk has been shown to decrease by up to two orders of magnitude in some cases, independent of FZ silicon manufacturer. High temperature (>1000 ℃) thermal treatments in an oxygen ambient have been shown to annihilate the point defects responsible for this instability in the bulk lifetime, however such processes are not readily available to all research groups, and thus are often omitted in sample processing. Czochralski (Cz) silicon wafers can offer greater thermal stability, as they do not possess the same point defects created in FZ silicon, however the bulk lifetime in ‘as-received’ wafers can degrade/improve via other mechanisms, such as oxygen-related defects (thermal donors, oxygen precipitation) and metal impurities. No silicon material can offer complete thermal stability of the bulk lifetime, and this could partly explain the variability in the passivation results reported in the literature. Setting aside material quality, and assuming similar deposition conditions, the main parameters that control Al2O3 surface passivation are found in the post deposition annealing conditions, as identified in Table 1. For the examples listed in Table 1, all studies have used FZ silicon as their base material without considering thermal degradation (or improvement), and each study used different post-deposition annealing conditions to achieve maximum surface passivation.
In this work, we show how recombination at the silicon-Al2O3 interface can be controlled (and inhibited) in order to maximise its passivation potential for silicon based solar cells and electronic devices. We begin by examining the effective lifetime of Al2O3 passivated FZ and Cz silicon under varying annealing ambient and temperature. We then strip the Al2O3 films from selected samples and use a room temperature superacid-based surface passivation technique to accurately measure the bulk lifetime. We subject selected Al2O3 passivated samples to corona charging in order to separate chemical from field-effect passivation contributions as a function of annealing temperature and film thickness. We then utilise transmission electron microscopy and energy dispersive X-ray spectroscopy to analyse the composition and distribution of elements within the Al2O3 films as the annealing temperature is increased. Finally, we perform grazing incidence X-ray diffraction (XRD) to determine crystallisation changes in the films.
2. Experimental methods
Immediately following the wet chemical cleaning process, the samples were transferred to the load lock of a Veeco Fiji G2 system which was then subsequently evacuated to mitigate any unintentional oxide formation. 5–250 cycles of Al2O3 (0.7–35 nm assuming a growth rate of 0.13 nm ) were deposited by ALD at 200 ◦C using an O2 plasma source and trimethylaluminum precursor. The deposition was performed on both sides of the samples to achieve symmetrical structures. Following the Al2O3 depositions, the samples were annealed in a quartz tube furnace for 30 min in air at a specified annealing temperature. In some cases, silicon samples were annealed in a Surface Science Integration Solaris 100 rapid thermal processor (RTP) for 30 min at a specified annealing temperature and ambient (N2, forming gas or N2O). For this study, a 30 min anneal was chosen to ensure enough time was allowed to achieve maximum passivation, especially for lower annealing temperatures.
3. Results and discussion
Fig. 1 (a) plots the effective lifetime (at an excess carrier density, Δn = 1015 cm− 3 ) of ALD Al2O3 coated (~20 nm) FZ 2 Ω-cm n-type silicon materials after annealing at temperatures between 360─520 ◦C in an RTP furnace for 30 min. The data in Fig. 1a show an increase in effective lifetime with annealing temperature, peaking at a temperature of 460─480 ℃, and then rapidly declining with higher annealing temperatures. Notably, the annealing ambient (N2, forming gas or N2O) has no significant influence on the level of passivation achieved with annealing temperature implying the improvement in passivation post annealing is primarily governed by the elemental species within the film (e.g., hydrogen) rather than originating from external sources. While the optimisation study shown in Fig. 1 (a) is very important for understanding how to maximise surface passivation, interpreting the results can become difficult when considering the underlying bulk material, as this can also change with the annealing conditions used to activate the passivation. Recently it has been demonstrated that ‘as-received’ FZ silicon is thermally unstable, whereby the bulk lifetime can degrade by two orders of magnitude over the temperature region in which Al2O3 passivation is thermally activated due to nitrogen-vacancy defects. Therefore, to overcome this limitation, we have assessed the bulk lifetime of the samples used in Fig. 1 (a), by stripping their Al2O3 coatings and subsequently re-passivating their surfaces using a room temperature superacid passivation method, as shown by the orange squares. Although the effective lifetime of the re-passivated samples is lower than that for Al2O3 passivation, (in keeping with the reported SRVs for Al2O3 and superacid of ~ 0.5 cm/s and < 2 cm/s , respectively) the trend is still indicative of variations in the bulk lifetime. As shown by the orange squares in Fig. 1 (a), the bulk lifetime is stable up to an annealing temperature of 480 ℃, above which it starts to degrade. Therefore, at annealing temperatures > 480 ℃ the bulk lifetime strongly influences the effective lifetime, meaning limited information regarding the stability of Al2O3 passivation at higher annealing temperatures can be gained from these measurements.
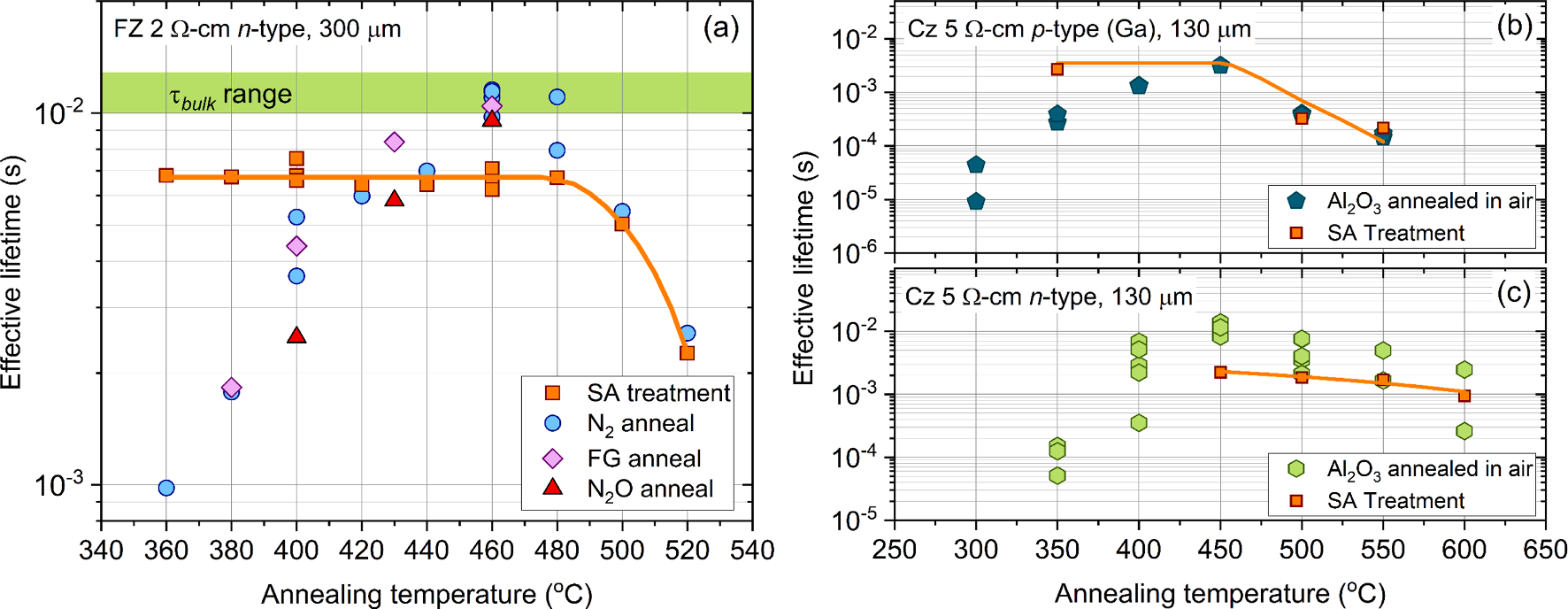
Fig. 1
From the minimum lifetime measurements presented in Fig. 2 (a) and 2 (b), we can separate the effects of chemical and field-effect passivation of the Al2O3 passivated samples when subject to various annealing temperatures. Fig. 2 (c) plots the amount of negative charge in the Al2O3 films with annealing temperature for both n- and p-type silicon samples. As should be expected, the negative charge in the Al2O3 films on n- and p-type silicon do not differ significantly, however their trends with annealing temperature show a slight monotonic increase in charge from − 4 × 1012 qcm− 2 at 350 ◦C to − 5.5 × 1012 qcm− 2 at 600 ℃.
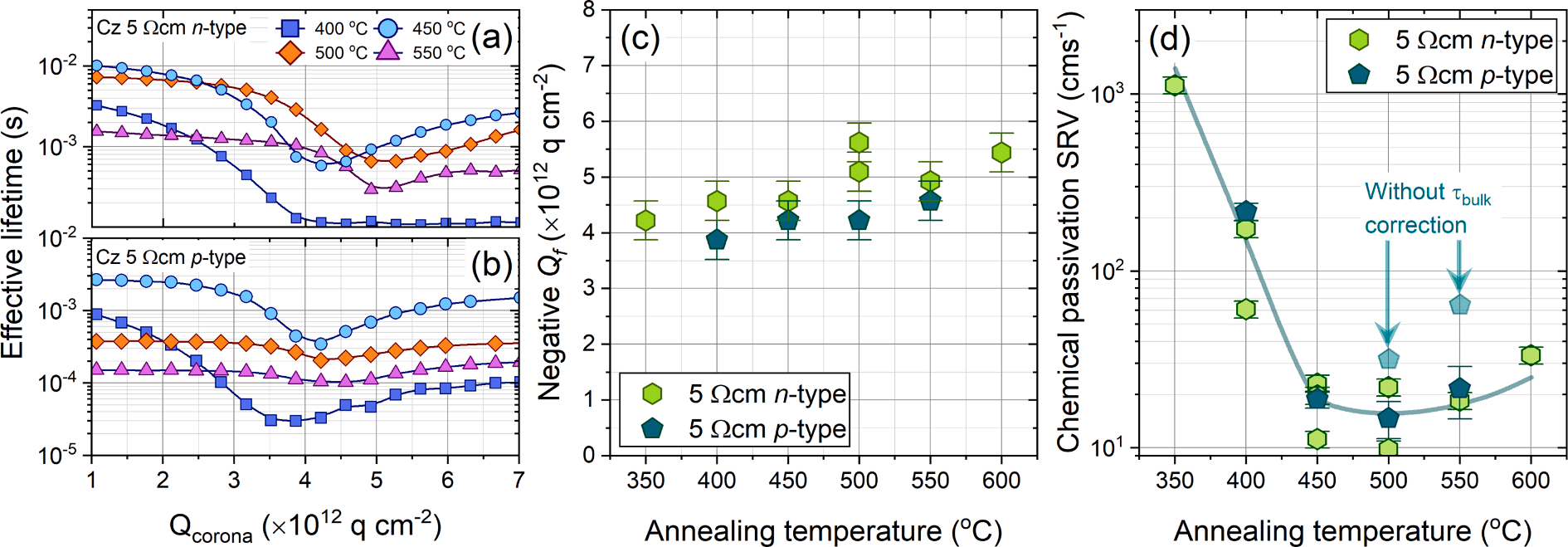
Fig. 2
Corona charge analysis demonstrated that Qf does not vary substantially with annealing temperature, as shown in Fig. 2 (b), hence we conclude that the trend in SPV observed in Fig. 3 (a) results from a reduction in Dit (and hence surface recombination) as annealing temperature increases, which is consistent with the chemical passivation results presented in Fig. 2 (d).Therefore, although it is difficult to ascertain the source of passivation, the trend shown in Fig. 2 (d) is consistent with a forming gas annealed thermal SiO2 layer whereby the additional source of passivation is hydrogen [35]. Thus, noting that plasma enhanced ALD Al2O3 contains ~ 3 at.% hydrogen at a deposition temperature of 200 ◦C, it is plausible that hydrogen is also a passivation source for Al2O3 on silicon.
Fig. 5 (g) and (h) show ADF-STEM images of ALD-grown Al2O3- coated (~20 nm) silicon samples annealed at temperatures of 200 ℃ and 500 ℃ for 30 min, respectively. Correspondingly, Fig. 5 (i) and (j) show cross-sectional intensity line scans from the ADF-STEM images in (g) and (h) respectively. In the as-deposited case, an oxide of ~ 5 nm thickness is present at the silicon interface, as shown in Fig. 5 (g) and inferred from (I). However upon annealing at temperatures ≥ 350 ℃ the oxide thickness is reduced (<2 nm in the case of a 500 ◦C anneal), as shown in Fig. 5 (h) and (j), with a slight compositional change due to the in-diffusion of Al, as demonstrated in Fig. 5 (c)-(f). Thus, unless ALD Al2O3 is annealed at temperatures ≥ 350 ℃, the interface oxide layer remains a dynamic layer in which thickness and composition can change depending on the processing/working temperature.
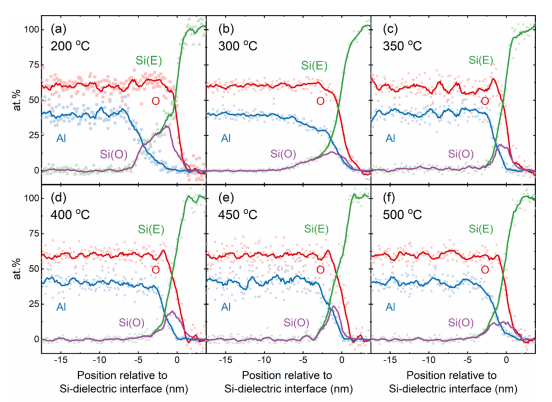
Fig. 5
4. Conclusion
In this work, we have conducted a thorough investigation of the mechanisms behind the activation temperature-dependent passivation quality of ALD Al2O3 films grown on n- and p-type silicon, separating bulk and surface recombination. We demonstrate that the ambient in which the Al2O3 films are annealed does not appear to have an influence on the level of passivation achieved. We also demonstrate tha maximum surface passivation is achieved with a post-deposition annealing temperature of ~ 450 ℃ for 30 min for both n- and p-type silicon. For higher annealing temperatures, the effective lifetime was found to decrease monotonically. However, upon removing the Al2O3 films and re-passivating the surface using a room temperature superacidbased technique, we show that this reduction in passivation is due to degradation of the bulk lifetime. The degrading bulk lifetime dominated the overall effective lifetime, and would yield inaccurate results of the surface passivation quality if this were not understood. By accounting for the reduction in bulk lifetime, in conjunction with corona charging and Kelvin probe experiments, we were able to demonstrate that the chemical passivation of Al2O3 films is stable between annealing temperatures of 450─500 ◦C. In contrast, the negative charge within the films was found to vary with temperature, increasing from –4 × 1012 qcm− 2 at 350℃ to − 5.5 × 1012 qcm− 2 at 600 ℃. In conjunction with the thermal stability, we also examined the film thickness dependence on the chemical and field effect passivation. We found that films as thin as 3 nm can achieve maximum chemical passivation when annealed at 450 ℃, achieving a Dit of ~ 3 × 1010 eV-1cm− 2 . Thicker films show no further reduction in the Dit, whilst Qf remains constant at ~ 5 × 1012 qcm− 2 within the 2–30 nm range.