ABSTRACT
Efhiciently passivating germanium (Ge) surfaces is crucial to reduce the unwanted recombination current in high-performance devicesChemical surface ceaning is critical to remove surace contaminants and Ge oxides, ensuring effective surace passivation after dielectricdeposition. However, Ge oxides can rapidly regrow upon air exposure. To understand the surface evolution ater wet cleaning, we present acomprehensive study comparing HF and HCl deoxidation steps on ptype Ge surfaces and monitor the surace as a function of air exposuretime. Distinct oxide regrowth dynamics are observed: HF-treated samples exhibit swift regrowth of all Ge oxide states, whereas HCl-treatedGe surfaces exhibit a lower concentration of low degrees of oxidation and slower or no regrowth of high oxide states even after 110 min ofair exposure. In addition, the presence of Ge-Cl bonds induces diferent oxidation dyamics compared to the Ge-0H bonds resulting fromHF deaning. This leads to varying surface electronic band structures, with HF-treated Gie exhibiting a stong positive band bending(+0 .20 eV). Conversely, HCl-treated samples display a lower band curvature (+0.07 eV, mostly due to the preence of Ge-Cl bonds on theGe surace. During air exposure, the increased GeO, coverage sienincantly reduces the band bending alter Hk, while a constant bangbending is observed after HCl. Finally, these factors induce a reduction in the surace recombination velocity after wet etching. Combiningboth chemical and field-induced passivation, HF-treated Ge without rinsing exceeds 800 us.
I. INTRODUCTION
Germanium (Ge) is used for many applications such as metal–oxide–semiconductor field-effect transistors (MOSFETs),free-standing membranes,IR lenses, biosensors,batteries,or multijunction solar cells. In most cases, high-performance device fabrication requires a proper understanding and control of the Ge surface passivation. Indeed, the potential of Ge is hampered by its high surface defect density. These defects lead to a high charge carrier recombination rate at the surface and can compromise the channel’s carrier mobility in MOSFET devices. In optoelectronic devices, such as solar cells and lasers, the electronic states induced by surface defects can act as nonradiative recombination centers for minority charge carriers and, therefore, decrease the device conversion efficiency.To counteract this detrimental surface impact on the final device’s performances, various passivation paths have been developed based on dielectric layer deposition.Although many of these studies were more interested in determining and optimizing the deposition parameters and the dielectric layers to obtain high passivation quality, some authors have mentioned the importance of the cleaning step performed prior to the deposition of the dielectric layer on the passivation performances.Ge wafers are traditionally exposed to wet cleaning to etch the native Ge oxide and prevent oxide regrowth. However, unlike on silicon, the peroxide-based solution cannot be used with Ge due to their high etching rates.From this observation, several cleaning techniques have been developed using water, ammonium hydroxide (NH4OH),thiols (–SH),or organic compounds.The cleaning step based on halogenated acid etchants (mostly, HF and HCl)has rapidly become widespread in the Ge industry by analogy with Si industrial cleaning steps.
In this work, we present a comprehensive comparison between HF and HCl cleaning procedures on p-type Ge (100) surfaces and elucidate the modification of both the surface composition and the electronic band structure after these treatments by parallel angle-resolved x-ray photoelectron spectroscopy (AR-XPS) measurements. In addition, their evolutions with air exposure dependence are clarified. Finally, we use microwave-detected photoconductivity (MDP) measurements to study the influence of these halide treatments and their relative surface modifications on the minority carrier lifetime.
II. EXPERIMENT
A. Samples
P-type gallium-doped, 50 mm diameter Ge wafers with a resistivity of 5–15 Ω cm and (100) crystal orientation with a 6°miscut toward (111), as well as hydrofluoric acid (HF, 49 wt.%), hydrochloric acid (HCl, 37 wt.%), and ultrapure water (H2O), are used in this study. Ge samples received two treatments: either HF or HCl for 5 min. For HF-treated samples, a water rinse for 1 min has been added for safety reasons. After each cleaning procedure, the samples were dried by a nitrogen gun. To study the influence of oxide regrowth (the queue time), the samples were left in the ambient air in a cleanroom with 45% humidity and 21℃.
B. Material characterizations
The chemical composition as well as the surface band structure are investigated with parallel AR-XPS (Thermo Avantage Theta 300). x-ray photons are generated with a monochromatic Al Kα x-ray source (1486.6 eV). The angular mode uses eight angles regularly spaced between 23:75° and 76:25° , referred to as the normal of the wafer (i.e., collection angles). The angles of 23:75° and 76:25° correspond to photoelectrons escaping from the bulk and near-surface with a probed depth of about 8 and 2 nm, respectively. The XPS step size is 0.05 eV, the pass energy and dwell time are set at 60 eV and 500 ms, respectively. The Ar floodgun is a combined Thermo Fisher Scientific dual Ar ion and electron flood gun used to compensate for surface charging effects with minimal surface damage.
III. RESULTS AND DISCUSSION
A. Impact of wet treatment on the surface composition and band bending of Ge
Preliminary AR-XPS experiments are performed on an as-received Ge substrate with its native oxide, hereafter referred to as Ge as-received. Figure 1(a) shows a typical XPS Ge3d core-level spectrum recorded at a collection angle of 76:25°. This spectrum has been decomposed into five contributions corresponding to Ge–Ge bonds from the bulk Ge and GeOx bonds corresponding to the four Ge surface oxidation states: Ge1þ, Ge2þ, Ge3þ, and Ge4þ. The Ge3d peaks related to Ge–Ge and Ge4þ bonds are fitted with a doublet using a spin–orbit splitting of 0.6 eV and an area ratio between the d5=2 and d3=2 components of 0.66. Due to the energy splitting of the Ge3d orbital, the doublet of the GeOx (1≤χ≤3) peaks is not resolved, and these contributions are fitted with a single peak. The Ge3d5=2 peak associated with the bulk Ge–Ge bonds is located at 29.8 eV, while the peaks related to the Ge1þ, Ge2þ, Ge3þ, and Ge4þ oxidation states are shifted with respect to Ge3d5=2 to a higher binding energy by 0.8, 1.8, 2.6, and 3.4 eV, respectively.33 In addition to Ge, oxygen and carbon are detected on the Ge surface. The O1s peak [Fig. 1(b)] is fitted with two contributions located at 531.1 and 532.3 eV, which can be assigned to O–Ge–Ge and a combination of O–C and O–Ge–O (second neighbor effect) chemical environments,45 respectively, present in the native oxide and carbonaceous pollution. The reconstructed surface composition, calculated from the atomic concentration profile collected at various angles, is represented in Fig. 1(c). In addition to carbon contamination, the atomic concentration profile shows that the native oxide formed on the Ge surface is 1.5 nm thick (depth at which the atomic percentage of Ge Bulk is higher than 90%). The native oxide is mainly composed of Ge4þ except at the interface between the Ge oxide and Ge bulk, where the Ge1þ oxidation state dominates.
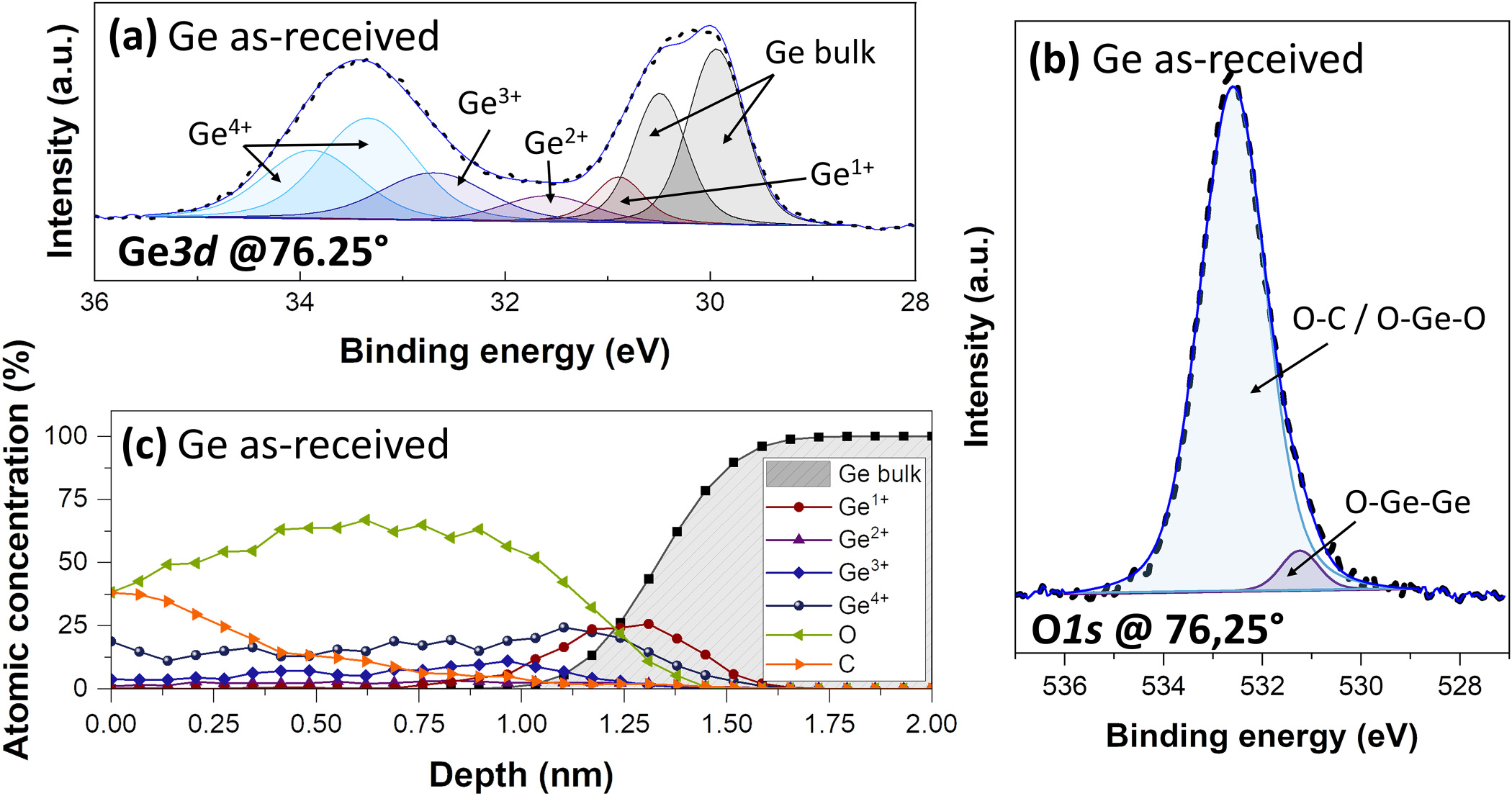
FIG. 1. XPS data for the as-received Ge sample. (a) Typical Ge3d peaks at a collection angle of 76:25° (better surface sensitivity). (b) O1s spectra at a collection angle of 76:25 . (c) Atomic concentration profile as a function of the depth.
The Ge substrates are immersed either in concentrated HF for 5 min followed by an H2O rinse for 1 min or in concentrated HCl for 5 min (without rinsing). XPS analyses are carried out 5 min after the wet dip. In both cases, the XPS analyses reveal a complete removal of Ge3þ- and Ge4þ-based compounds, as depicted in Figs. 2(a) and 2(b) for HF-treated and HCl-treated samples, respectively. This is also accompanied by a strong decrease in the O1s peak intensity [Fig. 2(c)]. The decrease is less in the case of HF treatment, certainly because water rinse induces a quick surface oxidation by the dissociative adsorption of H2O.After HF treatment, only traces of F-based compounds are detected [, 1%, Fig. 3(a)]. On the other side, after HCl treatment, we observe the appearance of a Ge–Cl peak with a chemical shift of 0.6 eV, in this case. The presence of this peak is confirmed by the Cl2p peaks located at 198.8 and 200.4 eV for the two spin–orbit peaks, 2p3=2 and 2p1=2, respectively [Fig. 3(b)]. In addition, a broad peak centered at 198.5 eV, in yellow in Fig. 3(b), is attributed to the Ge3s plasmon loss peak.
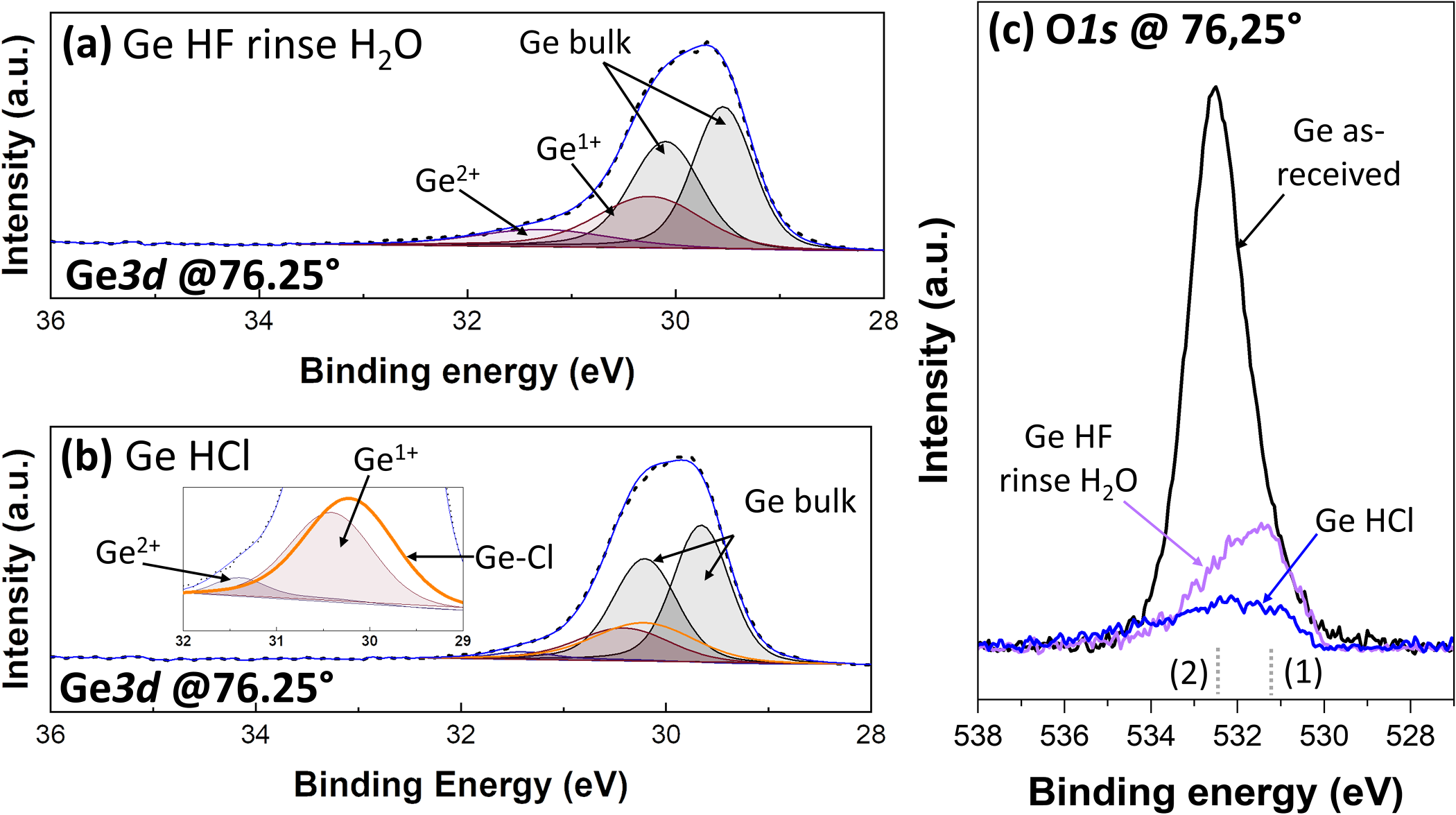
FIG. 2. XPS data after wet etching. (a) Typical Ge3d peaks at a collection angle of 76:25° after HF immersion for 5 min and water rinse. (b) Typical Ge3d peaks at a collection angle of 76:25° after HCl immersion for 5 min. (c) O1s spectra at a collection angle of 76:25° , the dotted lines (1) and (2) represent the peak positions of O–Ge–Ge and O–C/O–Ge–Ge, respectively.
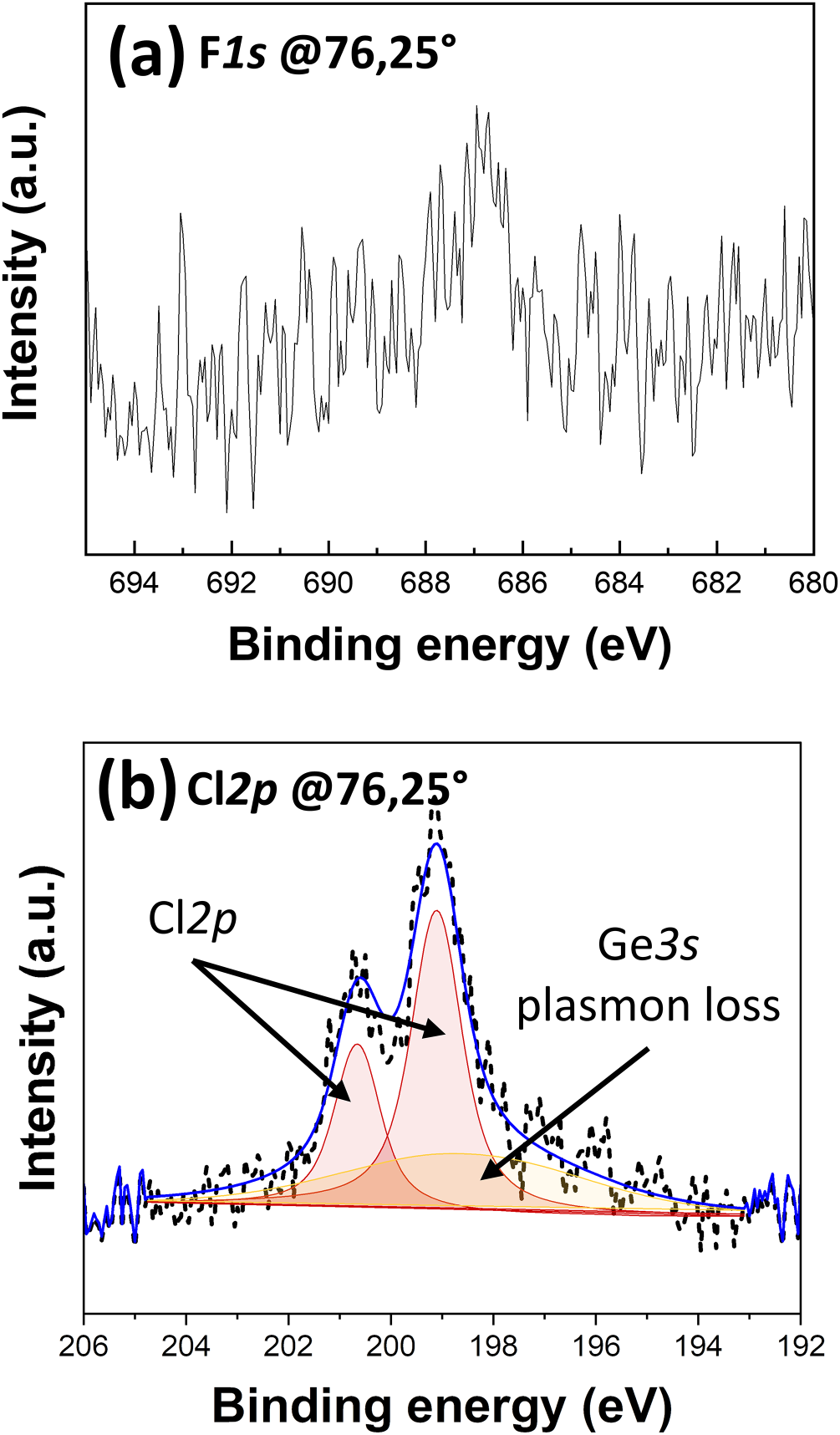
FIG. 3. (a) Typical F1s peaks at a collection angle of 76:25°after HF immersion for 5 min and water rinse. (b) Typical Cl2p peaks at a collection angle of 76:25°after HCl immersion for 5 min.
IV. CONCLUSIONS
The results presented in this study demonstrate that the cleaning procedure plays a critical role in the chemical composition, the surface band structure, the oxidation resistance, and the minority carrier lifetime of Ge. Our findings revealed varying oxide regrowth behaviors. For instance, Ge samples treated with HF exhibited a rapid regrowth of all degrees of oxidation, while those treated with HCl showed a reduced concentration of low degrees of oxidation following the cleaning and slower or negligible regrowth of high degrees of oxidation, even after 110 min of air exposure. This difference can be attributed to the presence of Ge–Cl bonds, which induce a distinct oxidation process compared to the Ge–OH bonds resulting from HF cleaning. As a consequence of these distinct surface compositions, Ge subjected to HF treatment with a water rinse displayed a pronounced positive band bending of þ0:20 eV. In contrast, HCl-treated samples exhibited a lower band curvature of þ0:07 eV, primarily due to the presence of Ge–Cl bonds on the Ge surface. Over the course of air exposure, an increase in the coverage of GeOx-based compounds led to a significant reduction in band bending after HF treatment, while HCl treatment maintained a consistent band bending. In summary, these factors collectively contributed to a reduction in the SRV after wet etching. By combining chemical and field-induced passivation, we achieved a minority carrier lifetime exceeding 800 μs in HF-treated germanium without rinsing.